Toward next generation of nanostructured materials
Our research is situated at the crossroads of chemistry, materials science, and engineering. We focus on the design, synthesis, and characterization of nanostructured materials—primarily nanoporous solids—to address critical societal challenges in energy efficiency, environmental remediation, and sustainable energy supply.
By controlling matter at the nanoscale, we engineer advanced nanomaterials designed to capture environmental pollutants, drive chemical transformations through heterogeneous catalysis, and harness solar energy for photocatalytic processes such as pollution degradation, water splitting, and CO2 reduction.
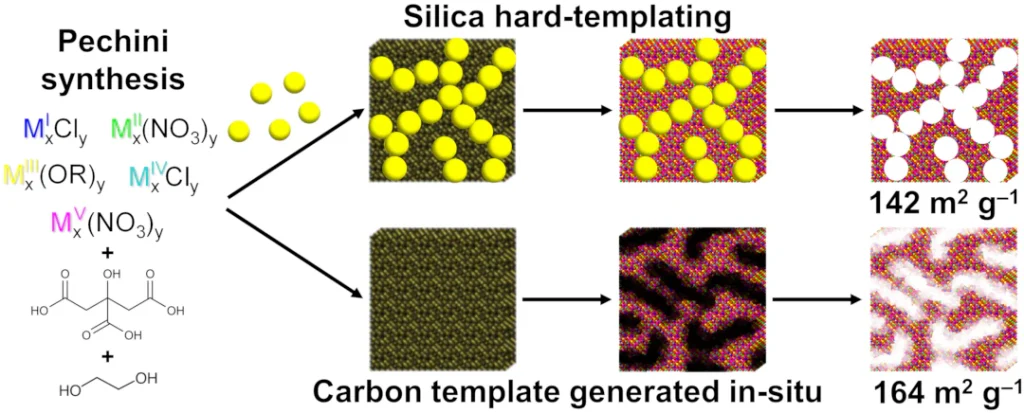
High-entropy oxides
High-entropy oxides (HEOs) leverage configurational entropy to stabilize complex crystal lattices containing five or more distinct metal cations. This structural complexity unlocks synergistic properties unattainable in simpler oxides. Our group develops novel synthesis pathways and elucidates structure-property relationships to deepen the fundamental understanding of these materials and accelerate their deployment in energy and environmental applications.
Image credit: Reprinted from Kandage, M. M.; Marszewski, M. Extension of Pechini Synthesis of High-Entropy Oxides: Preparation of Mesoporous High-Entropy Materials. Microporous Mesoporous Mater. 2026, 402, 113996., Copyright 2026, with permission from Elsevier.
Non-hydrolytic sol-gel synthesis
A major pillar of our lab is the development of new strategies for non-hydrolytic sol-gel (NHSG) synthesis of mesoporous transition metal oxides. Unlike traditional aqueous methods, NHSG synthesis provides superior control in the synthesis of transition and mixed metal oxides. We utilize it to engineer crystalline mesoporous architectures with high surface areas and ordered, uniform mesoporosity. These frameworks are designed to maximize the efficiency of a variety of heterogeneous and photocatalytic processes that rely on precisely engineered metal oxide surfaces.
Image credit: Copyright © 2012–2026 Michal Marszewski.

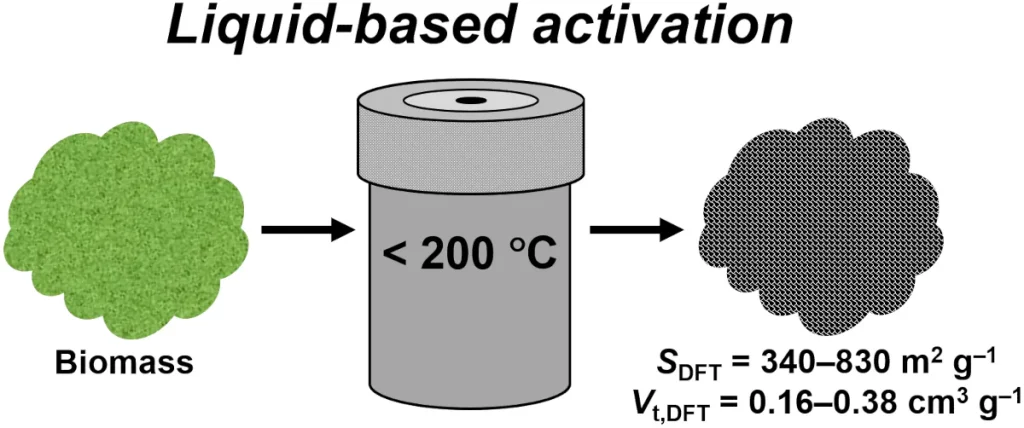
Bio-based and biomimetic materials
In line with green chemistry principles, we investigate the use of waste biomass and plant-based precursors to create functional materials, including activated carbons and biochars. Additionally, we explore the use of natural structures as templates for the synthesis of biomimetic metal oxides with complex, hierarchical architectures.
Image credit: Reprinted with permission from Bamidele, R.; Marszewski, M. Carbonization and Activation of Lignocellulosic Biomass in Liquid Phase at Sub-200 °C Temperatures. ACS Sustain. Chem. Eng. 2025, 13 (7), 3010–3021. Copyright 2025 American Chemical Society.